Official figures issued this summer highlighted the increasing rates of Type 2 diabetes in the UK, and the growing share of the NHS budget which it consumes. And since the poorest people are twice as likely to suffer Type 2 diabetes and its complications, this is also an issue which should concern all of us.
The figures, I’d like to suggest, are just the latest warning sign of a continuing public health disaster which began in 1984.
This was the year in which the Department of Health issued the first-ever governmental dietary guidelines for the population at large (not counting the advice issued during World War 2 on how to make the best of rationing).
Those 1984 guidelines (still promulgated in various forms today) were based on very shaky evidence – although I am not sure it even justifies the label of evidence; a better description might be ‘unproven hypothesis’ – which has fuelled both an obesity and diabetes epidemic. This hypothesis – that saturated fat causes heart disease – led to the advice that we should all fill up instead on carbohydrates such as pasta, bread and potatoes.
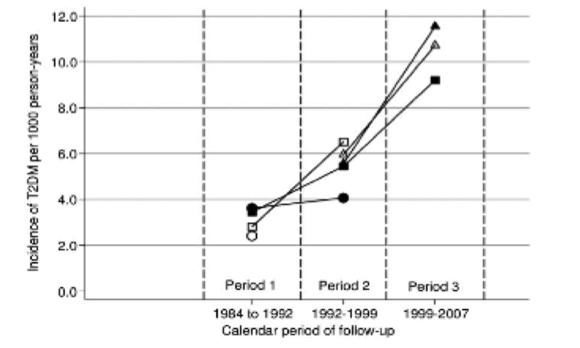
Just look at what happened to rates of Type 2 diabetes since then.
Figure 1: Trend over time (1984 – 2007) in incidence of T2 Diabetes by age group.
Key:❍ = age 45-49 years; ● = age 50-54 years; ❏ = age 55-59 years; ■ = aged 60-64 years;
▲ = aged 65-69 years; ▲ = aged 70-74 years. (Hardoon et al, 2010)
Ok, correlation is not causation – but is it just a co-incidence?
Most observers agree that the development of Type 2 diabetes is linked to an unhealthy lifestyle, including poor diet. And the NHS seems to be flailing about helplessly in the face of this problem; partly, of course, because it has to concentrate on fixing people when things go wrong, rather than helping us to stay well; but also, I suggest, because it still seems wedded to questionable theories about what constitutes a healthy diet – and individuals within the system are understandably afraid of taking up a career-limiting stand against current orthodoxy.
Perhaps they know what happened to Professor John Yudkin, the British scientist who rashly proposed in the 1970s that it was sugar, not fat, that was the dietary culprit in obesity and heart disease.
Start with food
Because it’s so much easier for us to live unhealthy lives, I’m not arguing that there is one simple fix. But since what we eat every day can have such profound impact on our health, surely food is one of the best places to start?
And one of the worst places to start is the NHS Eatwell Guide.
When I began studying for an MSc in Public Health Nutrition and Weight Management almost 10 years ago, whilst also working in the NHS, it became clear to me that I could not reconcile the dietary advice I could see being handed out to NHS patients with the evidence I was coming across in the science journals I had access to in the University library.
Research into low-carbohydrate diets, sparked by the popularity of the Atkins Diet, was consistently showing they were effective not only for weight loss, but for improved glycemic control in people with Type 2 diabetes, and a reduction in unhealthy blood fats and inflammation markers (or, in plain English, lower triglycerides and C-reactive protein).
The Eatwell Plate – designed by the food industry for wealth, not health
With my MSc under my belt, I could not in all conscience work in a nutrition-related role in the NHS where I would have to ‘toe the party line’ as far as the Eatwell Plate was concerned. This was a pictorial representation of government dietary advice, first issued in 2006, but the contents were simply a reinforcement of the 1980s advice to stuff ourselves with starchy foods).
Over 60% of the foods on the Eatwell Plate contained forms of sugar (glucose-fructose, fructose-glucose, lactose and long-chain starches which break down into glucose): bread, rice, potatoes, pasta, cornflakes, milk products, tinned baked beans, cakes, biscuits, chocolate, sweetened yoghurt, fruit juice, cola and fruit. Eat well? Really?
The Plate and guidelines were revised this summer with a call for a reduction of free sugars in the diet, but it still contains pictures of crisps, chocolate, cakes and ice-cream.
An editorial in the British Journal Of Sports Medicine said it was ‘designed by the food industry for wealth, not health’.
To compound the problem, the NHS still advises that we should ”Base meals on potatoes, bread, rice, pasta or other starchy carbohydrates. Choose wholegrain where possible Yet recent research indicates that wholegrain wheat is in fact no ‘healthier’ than white flour, since most modern wheat is a new hybrid which contains high levels of a highly-digestible form of starch – 70% carbohydrate by weight – called amylopectin A.
In healthy, slender volunteers, two medium-sized slices of wholewheat bread increased blood sugar by 30 mg/dl (from 93 to 123 mg/dl), no different from white bread. In people with diabetes, both white and wholegrain bread increased blood sugar 70 to 120 mg/dl over starting levels (Davis, 2011).
Far from helping us to ‘feel fuller longer’, wholegrain bread triggers a rise in blood sugar and higher levels of circulating insulin, followed by a sharp drop in blood sugar levels and increased hunger.
The NHS response to the growing body (you might almost call it obese) of evidence on the effectiveness of low carbohydrate patterns of eating is to stick its fingers in its ears and shout ‘La La La – can’t hear you.’ (see end of this article, a roundup of the robust evidence available to the NHS before it revised the Eatwell Plate this year).
Instead, the NHS advises: “Low-carbohydrate (low-carb) diets … tend to be high in fat, and eating a high-fat diet (especially saturated fat from foods such as meat, cheese, butter and cakes) could increase your risk of heart disease.” (www.nhs.uk).
Let’s deconstruct this a little…..
The advice does not specify what ‘high’ is, or what proportion of the diet should be made up of fat.
It suggests in a vague sort of way that a diet ‘rich’ in saturated fats could increase your risk of heart disease.
Saturated fat and heart disease
Six years ago, the Journal of the American Medical Association, having reviewed the evidence, stated that ‘saturated fat bears little relation to heart disease within most prevailing dietary patterns’ (JAMA, 2010). I sent a copy of this paper to NICE and asked if it would consider reviewing its advice in the light of the this information; and also if it could point me to the research on which its current advice (that saturated fat is dangerous) is based. It could not.
It’s not just the USA which is tiptoe-ing away from the diet-heart hypothesis about saturated fat and cholesterol. Several other studies, including major epidemiological reviews, meta-analyses of intervention studies and an Expert Report by the World Health Organisation, have failed to find any link between intake of saturated fat and heart disease (Mente et al, 2009, Siri-Tarino et al, 2010, Skeaff et al, 2009).
Low fat diets and heart disease
A Cochrane meta-analysis of 48 studies found that low-fat diets do not lower the risk of cardiovascular events or overall mortality (Cooper et al, 2011). In the two decades after World War 2, USA consumption of vegetable fat doubled while that of animal fat dropped by over 12%, yet heart disease rates soared (Taubes, 2007).
Equating meat and cakes
In warning people to avoid ‘low carbohydrate diets’, the NHS advice groups together meat, cheese, butter and cakes, which are very different types of food and contain very different macronutrients but which are all identified as ‘culprits’ because they contain saturated fat. Yet each food has a unique metabolic fate in the body and can affect health in very different ways.
For example, unless home-made, cakes are generally made with processed vegetable oil, and very often, this will be hydrogenated vegetable oil (trans fatty acids). This type of fat, although not a saturated fat, is now known to raise the risk of heart disease and stroke and increase levels of circulating LDL (Sun et al., 2007). Trans fats are also linked to an increased risk of Type 2 diabetes in women (Salmeron et al., 2001).
Industrially-produced cakes usually contain processed grains such as white flour (which will cause spikes in blood sugar). If the sugar in cakes is cane sugar (increasingly rare nowadays), it will also cause a rapid rise in blood sugar, reinforcing the impact of the processed white flour. More commonly in processed foods, the sweetener is high-fructose corn syrup, which, while it does not impact on blood sugar in the way that simple sugars do, is now being linked with non-alcoholic fatty liver disease. Also, since it does not provoke an insulin response in the way that cane sugar does, it does not stimulate the satiety signals which might limit the appetite (Johnson & Gower, 2009).
However, the ‘culprit’ in cakes – according to this advice – would be ‘saturated fat’.
As far as meat is concerned, if it is from grass-fed animals or non-factory-farmed poultry fed a natural diet which includes insects, worms and wild plants, then most of the fat in the meat will be monounsaturated fat, and rich in Omega-3 fatty acids, an essential dietary fat and a known anti-inflammatory (Maroon, 2006). If the meat is from grain-fed animals, then the meat is likely to be higher in Omega 6 fats, which can be deleterious to health if in a high ratio to Omega 3 intakes. (Simopoulous, 2002). A healthy ratio between Omega 3 and Omega 6 is no more than 2:1.
Margarine, promoted as a healthy alternative to butter, is, however, associated with heart disease (Gillman et al, 1997). For each teaspoon of margarine consumed a day, risk of heart disease was raised by 10% (Willett et al, 1993).
Food industry compliance
Of course, most ordinary people have probably never seen the Eatwell Plate – but the food industry loved it and sprang into action. We now see the consequences of it every day in the so-called foods which line our supermarket shelves, emblazoned with healthy-sounding labels like ‘wholegrain’ and ‘low fat’ (but stuffed with sugar, processed vegetable oils and salt to make them taste of something other than wet cardboard).
The 2010 position statement in the Journal of the American Medical Association (A Time For Food) criticises the nutrient-based official healthy eating advice of the last 40 years. (Of course, the USA started down this path a decade earlier than the UK and its ‘diabesity’ epidemic is, by a spooky co-incidence, about 10 years ahead of us.)
The JAMA paper goes on to say that this approach has fostered dietary practices that defy common sense, and allows highly-processed foods to be marketed as ‘healthy’ because they have replaced saturated fat with refined carbohydrates. It notes that the proportion of total energy from dietary fat appears largely unrelated to risk of cardiovascular disease, cancer, diabetes, or obesity. It also states that carbohydrate is a nutrient for which humans have no absolute requirement.
The paper concludes with calling for a major shift ‘to novel, evidence-based strategies (my emphasis) in which ‘foods’ [rather than individual nutrients] comprise the principal dietary targets.’
So how about it NHS?
Since in my view the roots of this dietary disaster lie in 1984, it seems appropriate to sign off with George Orwell, who presciently noted:
We may find in the long run that tinned food is a deadlier weapon than the machine-gun.
*****************************************
A sample of the evidence that was available before the NHS revised the Eatwell Plate this year
Systematic reviews
A number of metanalyses have found that the low-Glycaemic Index diet is effective for glycaemic control.
Thomas & Elliott’s (2010) looked at Randomised Control Trials from 1950 to 2009. Criteria for inclusion included randomised controlled trials (RCTs) lasting four weeks or longer; and comparisons between a low Glycaemic Index diet and a higher Glycaemic Index diet for people with diabetes. The standard measure for blood sugar (glycaemic) control is the level of glycated haemoglobin (HbAIc) over the previous three months. In these studies, HbA1c measures were used as the primary outcome of interest, with adverse effects, insulin action and quality of life as secondary outcomes of interest. Twelve papers met the inclusion criteria (612 participants).
Compared with people on the higher GI diets, there was a significant decrease in % HbA1c levels in people on low-Glycaemic Index diets, indicating improved glycaemic control in the low-Glycaemic Index group (WMD 20·4% HbA1c, 95% CI 20·7, 20·2, P1⁄40·001).There were also fewer episodes of hypoglycaemia, significantly higher insulin sensitivity, improved quality of life and compliance with the type of diet. In a study where medication needed adjusting, ‘significantly less medication was required in people with Type 2 diabetes on the low-Glycaemic Index diet than in those on the American Diabetes Association-recommended diet to achieve equivalent control of HbA1cs levels.’
The paper concluded that lowering the Glycaemic Index of the diet ‘appears to be an effective method to improve glycaemic control in diabetes and should be considered as part of the overall strategy of diabetes management.’
One trial (conducted in children) reported on quality of life, and found that it was significantly influenced by the type of diet: twice as many parents of those in the low-Glycaemic Index group than of those in the high-Glycaemic Index group stated that their children had no difficulties in selecting their own meals at the 12-month time point (51 v. 24%, P1⁄40·01). Also, almost twice as many parents of those in the low-Glycaemic Index group than of those in the high-GI group reported that diabetes never limited the type of family activities pursued (53 v. 27 %, P1⁄4 0·02).
In a meta-analysis, Kodama et al (2009) reviewed randomized trials that investigated two kinds of prescribed diets (a low-fat, high-carbohydrate [LFHC] diet and a high-fat, low-carbohydrate [HFLC] diet); the diets were isocaloric with similar protein intake. Nineteen studies (with 306 patients) met the inclusion criteria.
The LFHC diet significantly increased fasting insulin and triglycerides by 8% (p 0.02) and 13% (p 0.001), respectively, and lowered HDL cholesterol by 6% (p 0.001) compared with the HFLC diet.
The findings suggest that ‘replacing fat with carbohydrate could deteriorate insulin resistance while the adverse effect on triglycerides from the LFHC diet could be avoided by restricting energy intake to a degree sufficient for the attainment of weight reduction.’
A Cochrane Review (Moore et al, 2007) reviewed 36 articles reporting a total of 17 Randomised Control trials, of at least six months, involving 1,467 participants where dietary advice was the main intervention. Approaches included low-fat, high carbohydrate (LFHC), low-carbohydrate, high fat (LCHF), low-calorie (LC), (1,000 cal/d) and very-low calorie (VLC) (500 cal/d), and a modified fat diet. The study concluded that there are no high-quality data on the efficacy of dietary treatment of T2 diabetes, and there is an urgent need for well-designed studies examining a range of interventions.
Randomised controlled trials
A two-year study (Guldbrand & Nystrom, 2012) randomised 61 T2 diabetic patients into two groups, where they followed either a low-carbohydrate (high fat) diet (LCD) or a low-fat (LFD) diet. Both groups lost weight (approximately 4 kg on average), but after six months, the low-carb group had a clear improvement in glycaemic control, higher levels of HDL cholesterol and were able to lower Insulin doses by 30%. No such improvements were seen in the LFD group.
A Randomised control trial involving 215 newly-diagnosed and overweight Type 2 diabetic patients (Esposito et al, 2009) aged 30 to 75, over a period of 48 months, randomised subjects either to a low-carbohydrate Mediterranean (LCMD) diet, of <50gms of CHO/day; or to a low-fat diet (LFD). Participants and investigators were aware of the treatment assignment but assessors of the primary outcome (length of time to introduction of hyperglycaemic drug therapy) were blinded. Secondary outcomes were changes in weight, glycemic control and coronary risk factors.After four years, only 44% of patients in the Mediterranean-style diet group required treatment, compared with 70% in the low-fat diet group (absolute difference, 26.0 percentage points [95% CI, 31.1 to 20.1 percentage points]). Those on the Mediterranean-style diet lost more weight and experienced greater improvements in some glycemic control and coronary risk measures than those on the low-fat diet.
Similar results were observed in earlier RCTS (Dyson et al, 2007 and Samaha et al 2003) but the numbers were much smaller and the trials of shorter duration.
Bibliography
-
American Diabetes Association (2004).Weight Management Using Lifestyle Modification in the Prevention and Management of Type 2 Diabetes: Rationale and Strategies: a statement of the American Diabetes Association, the North American Association for the Study of Obesity, and the American Society for Clinical Nutrition. Diabetes Care, 27: 2067–2073
-
Arora, S. K., & McFarlane, S. I. (2005). The case for low carbohydrate diets in diabetes management. Nutrition & metabolism, 2(1), 16.
-
Assunção, M. L., Ferreira, H. S., dos Santos, A. F., Cabral, C. R., & Florêncio, T. M. M. T. (2009). Effects of Dietary Coconut Oil on the Biochemical and Anthropometric Profiles of Women Presenting Abdominal Obesity. Lipids, 44(7), 593–601.
-
Bravata, D. M., Sanders, L., Huang, J., Krumholz, H. M., Olkin, I., Gardner, C. D., & Bravata, D. M. (2003). Efficacy and safety of low-carbohydrate diets: a systematic review. JAMA: the journal of the American Medical Association, 289(14), 1837–1850.
-
British Broadcasting Corporation. (2011). The Food Programme. Trans Fats. Transmitted July 11. 2011.
-
Castañeda González, L. M., Gascón, M. B., & Cruz, A. J. (2011). Effects of low carbohydrate diets on weight and glycemic control among type 2 diabetes individuals: a systemic review of RCT greater than 12 weeks. Nutrición hospitalaria: Organo oficial de la Sociedad española de nutrición parenteral y enteral, 26(6), 1270–1276.
-
Cornier, M.-A., Dabelea, D., Hernandez, T. L., Lindstrom, R. C., Steig, A. J., Stob, N. R., Van Pelt, R. E., et al. (2008a). The Metabolic Syndrome. Endocrine Reviews, 29(7), 777–822.
-
Davidson, S., & Passmore, R. (1979). Human nutrition and dietetics (7th ed.), Edinburgh Press, Livingstone.
-
Dyson, P. A., Beatty, S., & Matthews, D. R. (2007). A low-carbohydrate diet is more effective in reducing body weight than healthy eating in both diabetic and non-diabetic subjects. Diabetic medicine: a journal of the British Diabetic Association, 24(12), 1430–1435.
-
Floegel, A., & Pischon, T. (2012). Low carbohydrate-high protein diets. BMJ, 344, e3801–e3801.
-
Gillman, M. W., Cupples, L. A., Gagnon, D., Millen, B. E., Ellison, R. C., & Castelli, W. P. (1997). Margarine intake and subsequent coronary heart disease in men. Epidemiology, 8(2), 144–149.
-
Greenberg, A. S., & Obin, M. S. (2006). Obesity and the role of adipose tissue in inflammation and metabolism. The American Journal of Clinical Nutrition, 83(2), 461S –465S.
-
Grootveld, M., Silwood, C. J. L., Addis, P., Claxson, A., Serra, B. B., & Viana, M. (2001). Health Effects of Heated Oils. Foodservice Research International, 13(1), 41–55.
-
Gross, L. S., Li, L., Ford, E. S., & Liu, S. (2004). Increased consumption of refined carbohydrates and the epidemic of type 2 diabetes in the United States: an ecologic assessment. The American journal of clinical nutrition, 79(5), 774–779.
-
Jenkins, D. J. A., Kendall, C. W. C., McKeown-Eyssen, G., Josse, R. G., Silverberg, J., Booth, G. L., Vidgen, E., et al. (2008). Effect of a low-glycemic index or a high-cereal fiber diet on type 2 diabetes: a randomized trial. JAMA: The Journal of the American Medical Association, 300(23), 2742–2753.
-
Johnson R.J., and Gower T. (2009). The Sugar Fix: The High-Fructose Fallout That is Making You Sick and Fat, Rodale. New York.
-
Karlström, B. E., Järvi, A. E., Byberg, L., Berglund, L. G., & Vessby, B. O. H. (2011). Fatty fish in the diet of patients with type 2 diabetes: comparison of the metabolic effects of foods rich in n-3 and n-6 fatty acids. The American Journal of Clinical Nutrition, 94(1), 26–33.
-
Kirk, J. K., Graves, D. E., Craven, T. E., Lipki=n, E. W., Austin, M., & Margolis, K. L. (2008). Restricted-carbohydrate diets in patients with type 2 diabetes: a meta-analysis. Journal of the American Dietetic Association, 108(1), 91–100.
-
Kodama, S., Saito, K., Tanaka, S., Maki, M., Yachi, Y., Sato, M., Sugawara, A., et al. (2009a). Influence of Fat and Carbohydrate Proportions on the Metabolic Profile in Patients With Type 2 Diabetes: A Meta-Analysis. Diabetes Care, 32(5), 959–965.
-
Kodama, S., Saito, K., Tanaka, S., Maki, M., Yachi, Y., Sato, M., Sugawara, A., et al. (2009b). Influence of Fat and Carbohydrate Proportions on the Metabolic Profile in Patients With Type 2 Diabetes: A Meta-Analysis. Diabetes Care, 32(5), 959–965.
-
Lindeberg, S., Jönsson, T., Granfeldt, Y., Borgstrand, E., Soffman, J., Sjöström, K., & Ahrén, B. (2007). A Palaeolithic diet improves glucose tolerance more than a Mediterranean-like diet in individuals with ischaemic heart disease. Diabetologia, 50(9), 1795–1807.
-
McAuley, K. (2006). Thematic review series: Patient-Oriented Research.: nutritional determinants of insulin resistance. The Journal of Lipid Research, 47(8), 1668–1676.
-
Mozaffarian, D., & Ludwig, D. S. (2010). Dietary guidelines in the 21st century—a time for food. The Journal of the American Medical Association, 304(6), 681–682.
-
Muraille E, Pajak B, Urbain J, Leo O. (1999). Carbohydrate-bearing cell surface receptors involved in innate immunity: interleukin-12 induction by mitogenic and nonmitogenic lectins. Cell Immunology. 10;191(1):1-9.
-
National Institute for Health and Clinical Excellence. (2011). Prevention of type 2 diabetes: reviewing mechanisms of successful interventions and translation of major trial evidence to practice.
-
National Institute for Health and Clinical Excellence.(2011). Preventing the progression of pre-diabetes to Type 2 diabetes in adults. Systematic review and meta-analysis of lifestyle, pharmacological and surgical interventions.
-
Nazroo, J. (1997) Genetic, cultural or socio-economic vulnerability? Explaining ethnic inequalities in health. Sociology of Health & Illness, 20, 5 pp. 710–730
-
Nestle, M. (2007). Food Politics: How The Food Industry Influences Nutrition and Health. University of California Press. Berkeley and Los Angeles.
-
NHS Information Centre: (2012). Prescribing for Diabetes in England: 2005/6 to 2011/12. retrieved from http://www.ic.nhs.uk/statistics-and-data-collections/primary-care/prescriptions/prescribing-for-diabetes-in-england-2005-6-to-2011-12
-
NHS: Diabetes Type 2: Causes: http://www.nhs.uk/Conditions/Diabetes-type2/Pages/Causes.aspx
-
Parillo, M., Rivellese, A. A., Ciardullo, A. V., Capaldo, B., Giacco, A., Genovese, S., & Riccardi, G. (1992). A high-monounsaturated-fat/low-carbohydrate diet improves peripheral insulin sensitivity in non-insulin-dependent diabetic patients. Metabolism: clinical and experimental, 41(12), 1373–1378.
-
Samaha, F. F., Iqbal, N., Seshadri, P., Chicano, K. L., Daily, D. A., McGrory, J., Williams, T., et al. (2003). A Low-Carbohydrate as Compared with a Low-Fat Diet in Severe Obesity. New England Journal of Medicine, 348(21), 2074–2081.
-
Salmeron, J., Hu, F. B., Manson, J. A. E., Stampfer, M. J., Colditz, G. A., Rimm, E. B., & Willett, W. C. (2001). Dietary fat intake and risk of type 2 diabetes in women. The American journal of clinical nutrition, 73(6), 1019–1026.
-
Sharma, S., Cade, J., Landman, J., & Cruickshank, J. K. (2002). Assessing the diet of the British African-Caribbean population: frequency of consumption of foods and food portion sizes. International journal of food sciences and nutrition, 53(5), 439–444.
-
Simopoulos, A. P. (2002). The importance of the ratio of omega-6/omega-3 essential fatty acids. Biomedicine & pharmacotherapy, 56(8), 365–379.
-
Sodhi, A., & Kesherwani, V. (2007). Production of TNF-alpha, IL-1beta, IL-12 and IFN-gamma in murine peritoneal macrophages on treatment with wheat germ agglutinin in vitro: involvement of tyrosine kinase pathways. Glycoconjugate journal, 24(9), 573–582.
-
Solinas, G., Vilcu, C., Neels, J. G., Bandyopadhyay, G. K., Luo, J.-L., Naugler, W., Grivennikov, S., et al. (2007). JNK1 in Hematopoietically Derived Cells Contributes to Diet-Induced Inflammation and Insulin Resistance without Affecting Obesity. Cell Metabolism, 6(5), 386–397. doi:10.1016/j.cmet.2007.09.011
-
Sun, Q., Ma, J., Campos, H., Hankinson, S. E., Manson, J. E., Stampfer, M. J., Rexrode, K. M., et al. (2007). A Prospective Study of Trans Fatty Acids in Erythrocytes and Risk of Coronary Heart Disease. Circulation, 115(14), 1858–1865.
-
Tappy, L. (2012). “Toxic” effects of sugar: should we be afraid of fructose? BMC Biology, 10(1), 42.
-
Taubes, G. (2007). The Diet Delusion. Ebury Publishing. New York.
-
Thomas, D. E., & Elliott, E. J. (2010a). The use of low-glycaemic index diets in diabetes control. British Journal of Nutrition, 104(06), 797–802.
-
Thomas, D. E., & Elliott, E. J. (2010b). The use of low-glycaemic index diets in diabetes control. British Journal of Nutrition, 104(06), 797–802.
-
Westman, E. C., Yancy, W. S., Mavropoulos, J. C., Marquart, M., & McDuffie, J. R. (2008). The effect of a low-carbohydrate, ketogenic diet versus a low-glycaemic index diet on glycaemic control in Type 2 diabetes mellitus. Nutrition & Metabolism, 5(1), 36.
-
Willett, W. (1993). Intake of trans fatty acids and risk of coronary heart disease among women. The Lancet, 341(8845), 581–585.
-
Xu, H., Barnes, G. T., Yang, Q., Tan, G., Yang, D., Chou, C. J., Sole, J., et al. (2003). Chronic inflammation in fat plays a crucial role in the development of obesity-related insulin resistance. Journal of Clinical Investigation, 112(12), 1821–1830.
Loren Grant is an independent nutritional therapist and researcher based in Chorlton, Manchester.
